|
Lauren Plumer, another spokesperson for the company, also emphasized J&J’s commitment to provide the vaccine to low- and middle-income nations in an email to PopSci. Jake Sargent, a spokesperson for Johnson & Johnson, told the New York Times that the company has millions of doses of the COVID-19 vaccine in inventory and will continue to meet its obligations to organizations that have put in orders on behalf of low-income nations. “There’s no doubt in my mind that it saved a lot of people and encouraged some people who may not have been willing to take the mRNA vaccine early on to at least start getting vaccinated.” “We have to give J&J credit where it’s due, that it fulfilled an important need very early in the pandemic,” Fuller says. These characteristics made the vaccine easier to distribute, particularly to people who lived in remote areas. Unlike mRNA vaccines, it doesn’t require ultra-cold storage conditions-and it’s given as a single shot, rather than two shots spaced several weeks apart. The J&J shot also comes with a few advantages of its own. However, all three are very effective at preventing serious illness from Omicron and other variants, Fuller notes. The J&J formulation has also been linked with rare but serious effects. The mRNA-based shots developed by Moderna and Pfizer have shown the most impressive results when it comes to preventing symptomatic COVID-19. “There’s no doubt in my mind that the J&J shot encourages some people who may not have been willing to take the mRNA vaccine early on to at least start getting vaccinated.” Deborah Fuller, vaccinologist at the University of Washington School of Medicine in Seattle Still others contain copies of the pathogen that have been weakened or “killed,” so they can’t make the recipient sick, much like the chickenpox and inactivated polio vaccines. Other vaccines contain proteins from the coronavirus, which is also how common immunizations against Hepatitis B and human papillomavirus work. 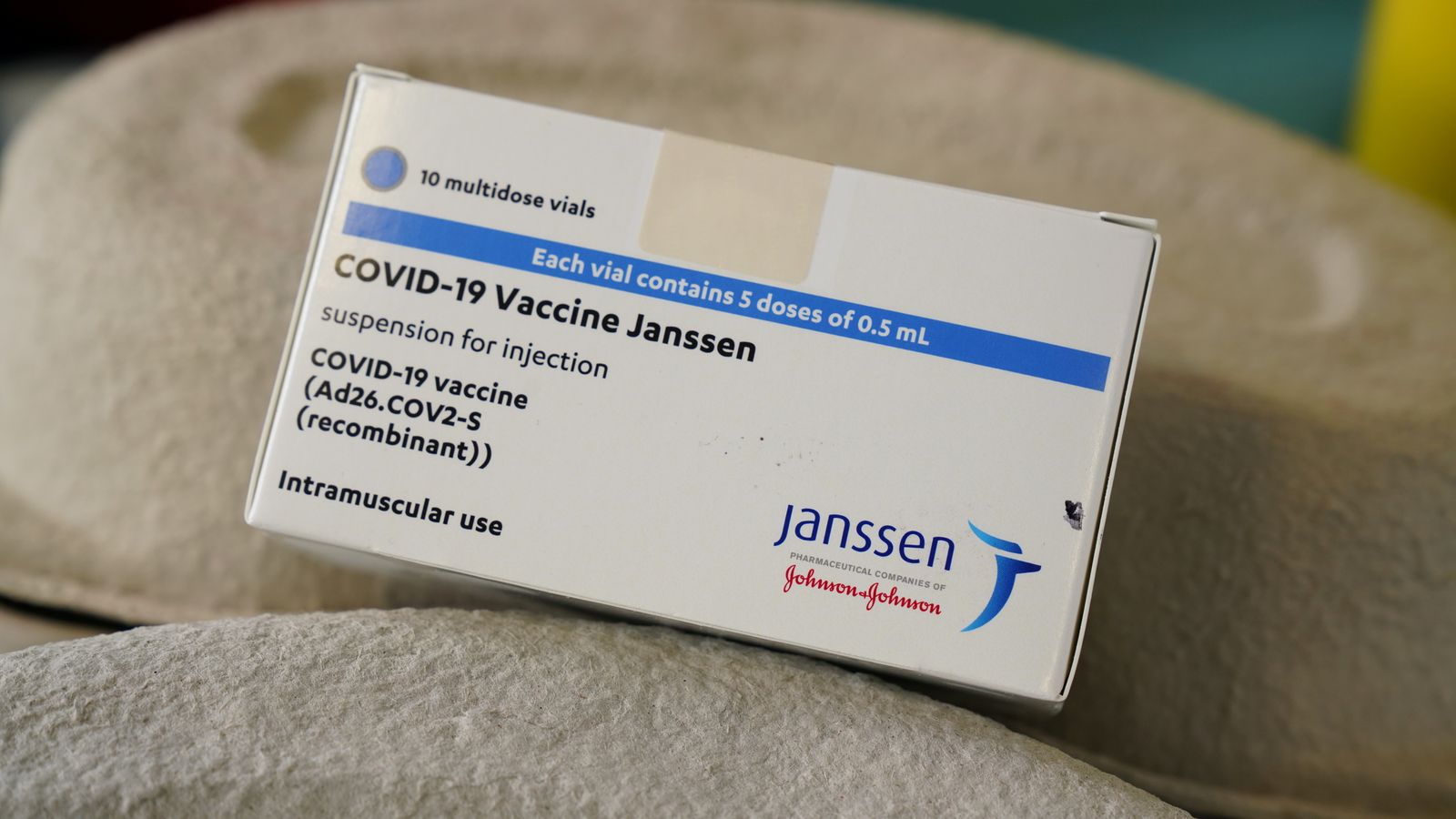
Other vaccines contain proteins from the coronavirus or copies of the pathogen that have been weakened or “killed” so they can’t make the recipient sick. Meanwhile, so-called viral vector vaccines, including those from J&J and the University of Oxford and AstraZeneca’s collaboration, use a different virus engineered to carry the coronavirus genes as a delivery vehicle. The recipient’s own cells use these instructions to build copies of the coronavirus protein, which then rouse the immune system without causing disease on their own. Some vaccines, including the ones produced by Moderna and Pfizer, contain genetic material that codes for the spike-shaped protein that helps the virus infect host cells. These vaccines use a variety of strategies to train the immune system to recognize and attack the novel coronavirus and its variants. More than two dozen different COVID-19 vaccines are in use around the world, and more than 100 are being tested in clinical trials. “What’s starting to happen with the J&J vaccine is that the unique part of its bubble is shrinking because other things are going to come into play.” “To stop the pandemic it’s not going to take one type of vaccine, but multiple different types of vaccine that fulfill different sorts of requirements,” Fuller says. What’s more, there are other kinds of vaccines out there with the same advantages as J&J’s, while being easier to mass produce. The number of COVID-19 shots that have received emergency authorization or full approval around the globe continues to grow. However, she added, the move is not necessarily cause for alarm. “The article came as somewhat of a surprise given that the world has not been vaccinated yet, and that was supposed to be the vaccine that had greater reach out there,” says Deborah Fuller, a vaccinologist at the University of Washington School of Medicine in Seattle. Nearly 40 percent of the world’s population is still unvaccinated, even as more than 92 million Americans have received a booster shot. 
But the pause raised concerns about accessibility for people in regions where few have been vaccinated against COVID-19. The plant, which is located in the Dutch city of Leiden, is expected to resume making the vaccines next month. On February 8, the New York Times reported that the only facility making usable batches of J&J’s COVID-19 vaccine shut down production and switched to manufacturing a drug for another respiratory virus late last year. However, many less wealthy nations like Mexico, Vietnam, and Zambia have relied on the J&J shot. 
Only 3.34 percent of shots administered in the country have used Johnson & Johnson’s formula. 
As the pandemic enters its third year, the COVID-19 vaccines developed by Moderna and Pfizer continue to dominate vaccination efforts in the US.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
